CHEMISTRY 3302 May 15, 1998
I. Molecular Properties (25 points)
Circle the one choice that fits the described property.
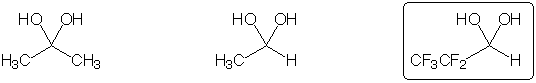
Hydrate found in greatest concentration at equilibrium in aqueous solution

Strongest acid
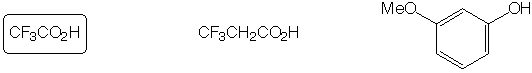
Slowest to react with H2SO4 / HNO3
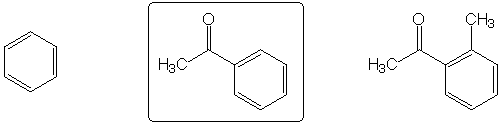
Highest boiling point
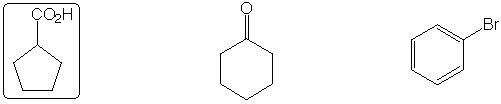
Fastest to react with MeO-
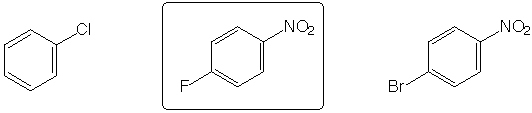
II. Chemical Transformations (28 points)
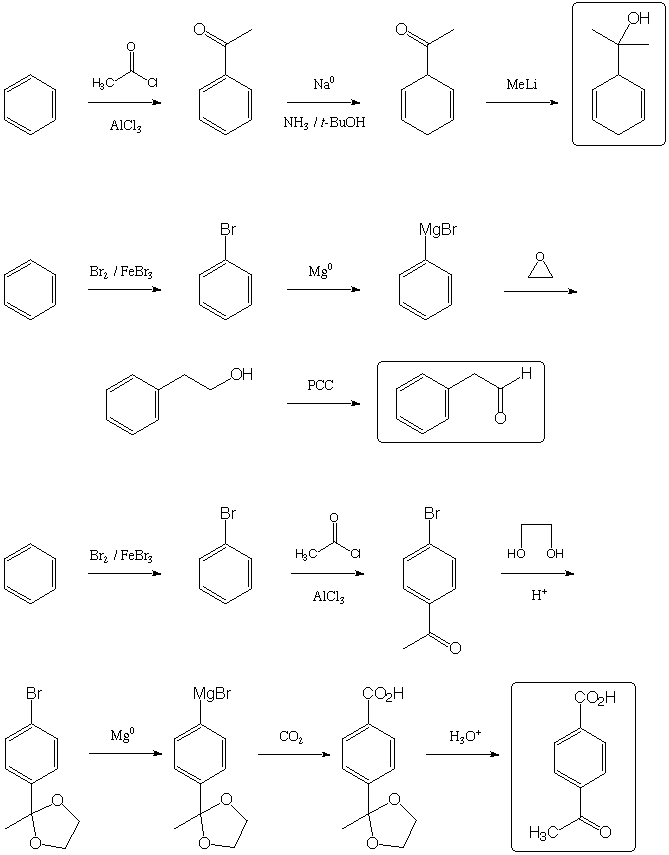
Fill in the boxes with the appropriate educts, reagents, or products.
A normal workup is assumed. Be sure to show stereochemistry if
necessary to distinguish isomers.
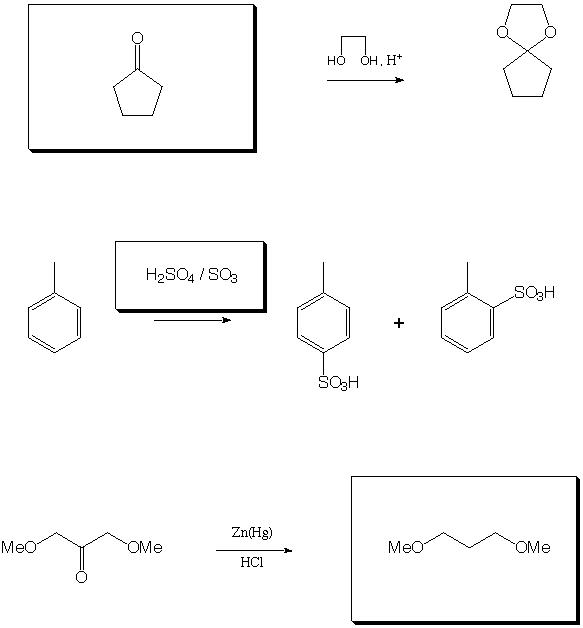
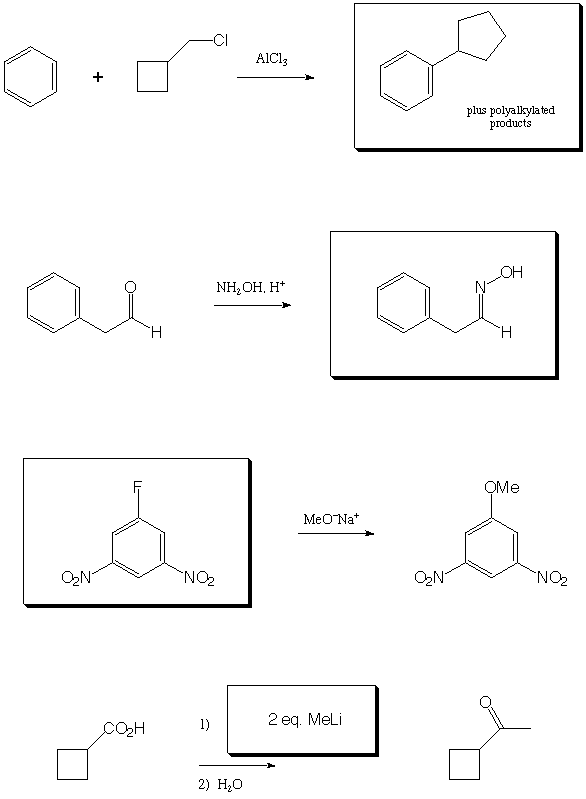
III. Organic Logic and Analysis (22 points)
a. Recall that the standard C=O stretching frequency (IR) is 1710 cm-1. Carbonyl groups on strained rings show higher frequency stretches. For instance, cyclopentanone has its C=O stretch at 1745 cm-1. Conjugation, on the other hand, lowers frequencies. Thus, 2-cyclopenten-1-one has a C=O stretching frequency of 1715 cm-1. Explain why conjugation lowers the C=O stretching frequency.
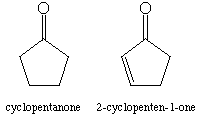
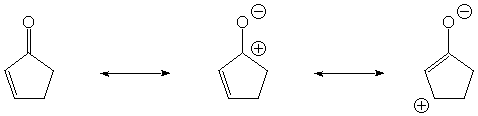
Conjugation provides an additional resonance structure (rightmost)
with CO single bond character.
b. 3Methyl-2-cyclopenten-1-one has a still lower frequency of 1705 cm-1. Why does methyl substitution further lower the frequency?
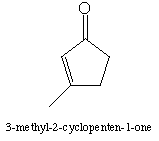
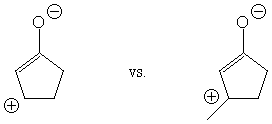
The weight of the charge-separated resonance structure (with
CO single bond character) is increased by methyl substitution,
since this stabilizes cationic charge at the substituted position.
c. (Hard) Cyclopentadienone, oddly enough, shows a C=O stretch at 1790 cm-1. Explain this higher frequency stretch. Extra ring strain in cyclopentadienone is not the answer.
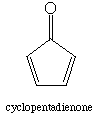
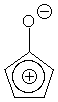
Delocalization of the positive charge now takes place in a cyclic ring of p orbitals, but the cyclopentadienyl cation is antiaromatic (4n pi electrons), so it is very unfavorable to have CO single bond character, and the C=O stretching frequency is correspondingly high.
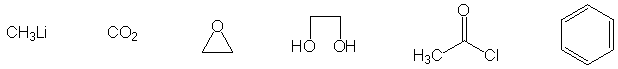
IV. Multistep Synthesis (25 points possible)
Complete the synthesis of any one of the target molecules.
The maximum points available per target is shown (harder targets
are worth more points). If you write down more than one synthesis,
indicate which one you want to be graded. For carbon atoms, you
may use only the carbon sources shown below. You may use any
other reagents you like, as long as they do not contribute
a carbon atom to the target. An extra page is attached if you
need more space.
Carbon sources:
Targets:
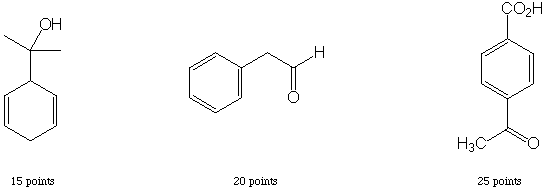
Other sequences than those shown below are possible. Note, however, that the Birch reduction for sequence 1 must occur before addition of methyllithium, or the cyclohexadiene will have the alternative double-bond regiochemistry. Also note that acetyl and carboxylate are meta directing groups, so in sequence 3 there is no shorter way to introduce these two groups para (although it is perfectly OK to acetylate and protect first and brominate second-in fact, it's probably better, since the bulk of the ketal will provide more para selectivity over ortho).