11/21/2008
Thermoreversible Micelle Shuttle Between an Ionic Liquid and Water
Recent research from the group of Professor Tim Lodge.
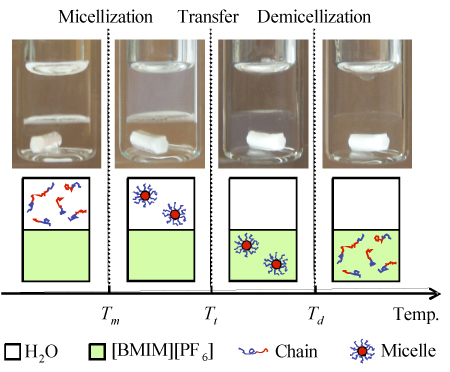
Micelles formed by self-assembly
of poly(ethylene oxide)-poly(N-isopropyl acrylamide) (PEO-PNIPAM) diblock
copolymers transfer, intact and reversibly, from water at room temperature to
the hydrophobic ionic liquid, [bmim][PF6], at elevated temperature.
This "micelle shuttle" should enable the facile delivery or extraction of
non-polar reagents to or from an ionic liquid reaction medium. Furthermore,
because the PNIPAM core blocks come out of aqueous solution near 34 °C, and
because they dissolve in the ionic liquid only above about 80 °C, this system
constitutes a completely automatic and thermoreversible
loading/transfer/delivery system. That is, the copolymers exist as free chains
in water, but micellize on heating above 34 °C. Further heating causes the
transfer into the ionic liquid, near 68 °C. Then, when heated above 80 °C, the
micelles dissociate, dispensing their cargo. On cooling, the cycle is reversed.
This work was carried out by
graduate student Zhifeng Bai, with help from postdoc Yiyong He and summer REU
student Nick Young, under NSF support. It is described in Macromolecules, 41,
6615-6617 (2008).
|