|
04/21/2010
An Old Plastic with a New Structure: Nanoporous
Linear Polyethylene
Recent research from the research group of Professor Marc Hillmyer.
Completely linear polyethylene (LPE) exhibits
incredible strength, durability, and chemical inertness, and thus finds use
in many aspects of our daily lives, from milk containers to car parts to snow
boards. New strategies to functionalize this ubiquitous plastic are continually
sought to improve the applicability to more technologically advanced products.
To this end, graduate students Louis Pitet and Mark Amendt working in Professor
Marc Hillmyers group combined two ring-opening polymerization mechanisms to
synthesize block polymers that were used to template nanoporous membranes.
The preparation of nanoporous LPE was recently reported in a communication
to the Journal of the American Chemical Society (J. Am. Chem. Soc. 2010,
ACS ASAP [http://dx.doi.org/10.1021/ja100985d]). The work was also recently
highlighted in a Chemical and Engineering News concentrate (April 12, 2010,
p. 29). The synthetic protocol relies on (1) the high functional group tolerance
of ruthenium mediated metathesis polymerization to form functionalized LPE
and (2) the sacrificial nature of the second component, polylactide, under
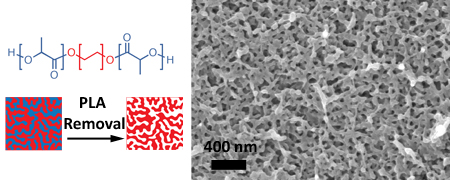
mildly basic conditions. The organization of block polymers into secondary
structures with segregated domains having nanoscopic dimensions (5-50
nm) is well-established. However, this particular system was designed to create
a microphase separated system that lacked a well-organized structure. This
strategy allows access to bicontinuous morphologies over large composition
windows. With respect to porous materials, this translates to easily tailored
porosity. Polylactide removal leaves a continuous LPE scaffold with interpenetrating
void space. The nanoporous LPE offers a ductile material impervious to degradation
in many harsh chemical environments that holds tremendous promise as, for example,
Li-ion battery separators.
|