|
02/17/2010
How many atoms can Th coordinate?
Recent research from the research group of Professor Laura Gagliardi.
The concept of coordination number
is extremely useful and widely employed to describe the local chemical
environments of atoms in matter. Originally defined by Alfred Werner in
1893, the coordination number is closely tied to many other important properties
such as atomic radius, molecular and electronic structure, and chemical
reactivity.
Very high Werner coordination numbers
are seen for metal complexes of the borohydride anion BH4. For
example, Zr(BH4)4, has a coordination numbers of twelve,
while Th(BH4)4 has coordination numbers of 14. No
complex of any kind, however, has been definitively shown to adopt a Werner
coordination number of 15.
A joint experimental and computational
work, respectively performed in the groups of Gregory S. Girolami, University
of Illinois and
Laura Gagliardi, University of Minnesota, has lead to the synthesis and
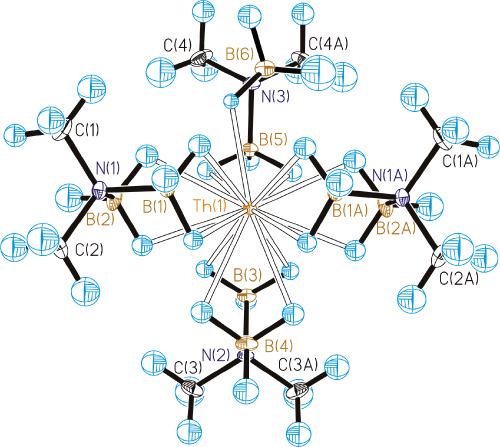
characterization of Th(H3BNMe2BH3)4,
the first 15-coordinate complex of such type. This work will appear soon
in Angew. Chemie Int. Ed. as a Communication.
Both the X-ray and neutron diffraction
results show that thorium forms bonds with fifteen hydrogen atoms; accordingly, this
is the first crystallographically characterized complex with a Werner coordination
number of 15. DFT calculations on the single Th(BH3NMe2BH3)4 unit
or a finite cluster of units do not reproduce the crystal structure. However,
imposing periodic boundary conditions gives a geometry for Th(BH3NMe2BH3)4 that
is in a very good agreement with that determined from experiment.
The present results set a new record – fifteen – for
the highest Werner coordination number seen to date in any form of matter.
This finding suggests that the discovery of metal complexes with Werner
coordination numbers equal to 16 should be possible with the right combination
of metal and ligands.
|