Main navigation | Main content
Despite its prominent position as the first intermediate in oxygen-activation mechanisms of many enzymes and synthetic complexes, well characterized nonheme iron-superoxo complexes are exceedingly rare. Such an elusive species has recently been identified in a collaborative effort among the groups of Professor Way-Zen Lee at National Taiwan Normal University, Professors Eckard Münck and Emile Bominaar at Carnegie Mellon University, and Professor Lawrence Que Jr. at the University of Minnesota, and this result has just been published online in the Journal of the American Chemical Society (DOI: 10.1021/ja504410s).
Also participating in this study were Ryan Chiang, Ph.D., and Po-Yi Li of Taiwan Normal, Scott Kleespies of the University of Minnesota, and Heather Stout and Katlyn Meier of Carnegie Mellon.
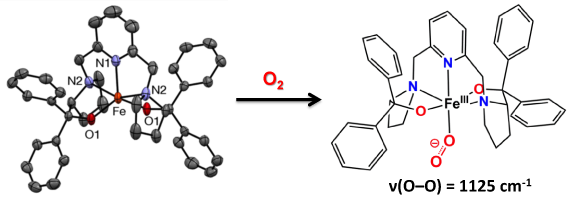
This first example of a mononuclear nonheme iron-superoxo complex was generated by exposure of the iron(II) complex to O2 at -80 °C in THF and characterized by a combination of UV-Vis, resonance Raman and Mössbauer spectroscopies. Properties of note are: a) the reversibility of O2 binding, b) an O-O stretching frequency of 1125 cm-1 that is consistent with assignment of the bound O2 as a superoxo ligand, and c) Mössbauer evidence for a high-spin iron(III) center. Thus, this reversible O2 adduct is paramagnetic, unlike its counterparts in heme chemistry. The consequences of this difference in magnetic properties on the ability of the bound O2 moiety to abstract an H-atom from substrate are being investigated.
This work has been supported by the Ministry of Science and Technology of Taiwan and the National Science Foundation.