

Main navigation | Main content
12/11/2015
Recent research from the research group of Professor
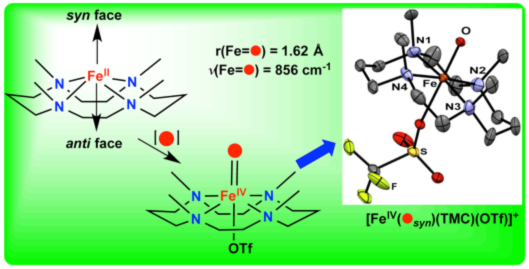
Twelve years ago, the first crystal structure of a synthetic nonheme oxoiron(IV) species [FeIV(O)(TMC)(NCMe)]2+ was reported by Lawrence Que Jr. and Wonwoo Nam1 (Science, 2003, 299, 1037), initiating an explosion of activity that has resulted in the characterization of more than 70 such complexes by a number of laboratories. Two additional structures of oxoiron(IV) complexes supported by closely related TMC-based ligands have been reported. In all three cases, the oxo atom is situated anti to the four methyl groups of the TMC macrocycle, suggesting a thermodynamic preference for this site. Recently, Jai Prakash, Ph.D, in the Que laboratory synthesized what appeared to be an isomer of the first [FeIV(O)(TMC)]2+ complex, and Gregory Rohde, Ph.D., solved its structure to demonstrate the anticipated syn binding of the oxo moiety. This result has just been reported in Inorganic Chemistry (Inorg. Chem. 2015, 54, 11055).
Crystals of [FeIV(Osyn)(TMC)(OTf)]+ were obtained at -80 ºC by recrystallization of the product from the reaction of FeII(TMC)(OTf)2 with the oxo-transfer reagent, 2-tBuSO2-C6H4IO in MeCN at 25 °C. Interestingly, when the same reaction was performed with C6H5IO as an oxidant, the anti isomer, [FeIV(Oanti)(TMC)(NCMe)]2+ was obtained, as previously reported. The syn isomer has the shortest FeIV=O bond (1.625(4) Å) found to date among all crystallographically characterized oxoiron(IV) complexes and exhibits UV-vis, IR, 1H-NMR and Mossbauer parameters distinct from those of the anti isomer. Further investigations are underway to determine whether structural differences give rise to different reactivities.
1Nam is a distinguished professor in the Department of Chemistry and Division of Molecular Life Sciences at Ewha Womans University in Seoul, Korea.